Due to its very low density, neon floats up into the atmosphere. 
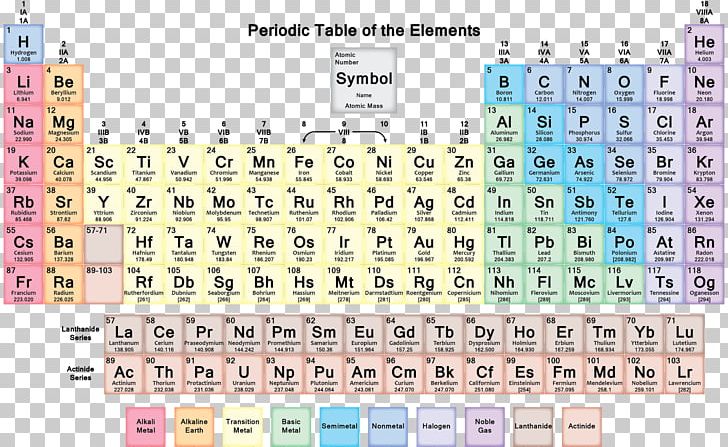
Neon gas is invisible to all human senses at room temperature: it’s invisible and has no odor or taste. For this reason, noble gases are used in situations that require a stable element to maintain a safe and constant environment. Elements in Group 18 are referred to as noble gases, called so because they are very unreactive and stable. Neon is the second element in Group 18 of the periodic table, appearing after helium. Neon’s name is derived from the Greek word “neos,” meaning “new.” The discoverer’s son actually proposed the name, and it’s possible he chose “new” because the color of neon’s glow was unlike anything that had been observed before. Neon is most famously known for its use in lighting applications like “neon signs” and lasers, but it is also used as a refrigerant. 
It is located on the right side of the periodic table with the other noble gases. Neon is a colorless, odorless, tasteless gas at room temperature and pressure. 1995 - 2023.“Neon” by Lestat has not been altered and is licensed under CC Attribution-Share Alike 3.0 Unported. If you need to cite this page, you can copy this text: This database focuses on the most common chemical compounds used in the home and industry. Molar mass calculations are explained and there is a JavaScript calculator to aid calculations. Molar Mass Calculations and Javascript Calculator.Introduces stoichiometry and explains the differences between molarity, molality and normality. Related ResourcesĪnswers many questions regarding the structure of atoms. Neon MenuĪ list of reference sources used to compile the data provided on our periodic table of elements can be found on the main periodic table page. It also forms an unstable hydrate, so it is not nearly as inert as one might think. Neon may also form ions in combination with other noble gases (NeAr, HeNe, Ne 2 and with hydrogen (NeH). Additional Notes: While it is inert, there have been reports of it combining with fluorine.Neon has also been used to make lightening arrestors, voltage detectors and TV tubes. 
Uses of Neon: In a vacuum tube, neon glows reddish orange, thus, the invention of the neon lights.Sources of Neon: It can be prepared by liquification of air and separated from other elements by fractional distillation.Note: this data represents naturally occuring levels of elements in the typical human, it DOES NOT represent recommended daily allowances. Specific Heat: 0.904J/gK Regulatory / Health.Description: Colorless, odorless, tasteless noble gas.Coefficient of lineal thermal expansion/K -1: N/A.Valence Electron Potential (-eV): Physical Properties of Neon.Electronegativity: N/A (Pauling) 4.84 (Allrod Rochow).Valence Electrons: 2s 2p 6 Electron Dot Model.Number of Neutrons (most common/stable nuclide): 10.Number of Electrons (with no charge): 10.Electrons per Energy Level: 2,8 Shell Model.Crystal Structure: Cubic face centered.Cross Section (Thermal Neutron Capture) σ a/ barns : 0.04.Series: Noble Gasses Neon's Name in Other Languages.Potential Parent Nuclides Overview of Neon.In addition technical terms are linked to their definitions and the menu contains links to related articles that are a great aid in one's studies. Common chemical compounds are also provided for many elements. Skip to site menu on this page Periodic Table of Elements Element Neon - NeĬomprehensive data on the chemical element Neon is provided on this page including scores of properties, element names in many languages, most known nuclides of Neon. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
